 /what-are-the-first-20-elements-608820-FINAL-5b758ab446e0fb002c67279a.png)
Cesium was discovered by Gustav Kirchhoff and Robert Bunsen (in 1860).The density of cesium is twice that of the density of water.Cesium is the 50th most abundant element found from the earth’s crust.The amount of cesium present in the earth’s crust is approximately 3 ppm by weight.Cesium was the first element that was discovered with a spectroscope.The name cesium was derived from the Latin word “caesius”, which means sky blue.Interesting facts about indium element are mentioned below. So the last electron of cesium enters the s-subshell or s-orbital. The simple answer: The elements will lie in the s, p, d or f block will completely depend upon the subshell in which the last electron will enter.įor example the electron configuration of cesium is 6s 1. How can you determine the blocks-wise position of elements? Gustav Kirchhoff and Robert Bunsen (in 1860) CAS number īefore knowing this reason, first of all I want to ask you a simple question. Protons 55 Neutrons 78 Electrons 55 Symbol Cs Atomic massĢ, 8, 18, 18, 8, 1 Electronic configuration 6s 1 Atomic radiusģ43 picometers (van der Waals radius) Valence electronsġ 1st Ionization energy 3.894 eV ElectronegativityīCC (Body centered cubic) Melting point 301.7 K or 28.5 ☌ or 83.3 ☏ Boiling point 944 K or 671 ☌ or 1240 ☏ Density 1.88 g/cm 3 Main isotope 133Cs Who discovered Cesium and when? Silvery pale gold State (at STP) Solid Position in Periodic table 
Let’s dive right into it! Cesium Element (Cs) Information Appearance So if you want to know anything about Cesium element, then this guide is for you. In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Cesium element in Periodic table.) Therefore, there are various non-equivalent definitions of atomic radius.This is a SUPER easy guide on Cesium element. However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space. 
The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. It must be noted, atoms lack a well-defined outer boundary. The atomic radius of Meitnerium atom is 129pm (covalent radius). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Meitnerium are 274, 276, 278, 282. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. 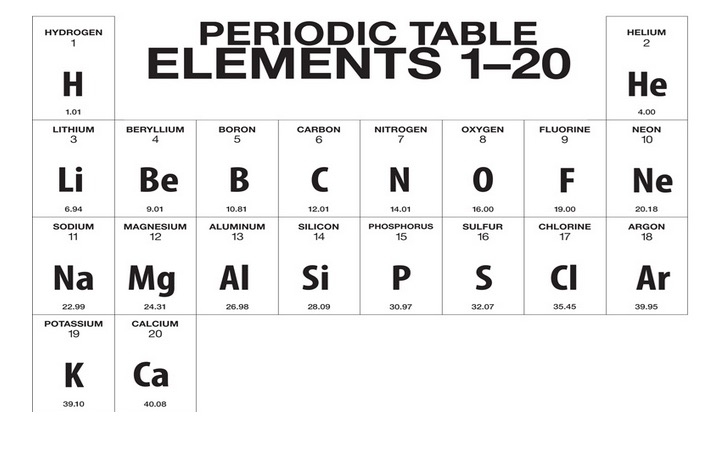
The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Meitnerium is a chemical element with atomic number 109 which means there are 109 protons in its nucleus. Atomic Number – Protons, Electrons and Neutrons in Meitnerium 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
